 
It is most often found in Stibnite (is a sulfide mineral with the formula, Sb 2S 3) Sometimes called antimonite. Abundance of AntimonyĪntimony is not an abundant element, but is found in over 100 mineral species.Īntimony can be found as the native metal. Inhalation of antimony trioxide is considered harmful and suspected of causing cancer. Pure antimony used in electronics industry to make certain types of semiconductor devices, such as infrared detector, diodes and hall-effect devices.Ī Antimony is alloyed with lead to improve their hardness and mechanical strength.Ī Antimony alloys are used in Batteries, antifriction alloys (such as Babbitt metal), type metal (linotype printing machines), electrical cable sheathing, small arms and tracer bullets.Īntimony is mainly used in the trioxide for make flame-proofing compounds, paint, glass art, and as an opacifier (Titanium dioxide, TiO 2) in enamel.īiological role: Antimony and many of its compounds are toxic. Naming: Greek: anti (not) monos (alone) Symbol for Latin stibium The lower-grade ores are reduced in blast furnaces while the higher-grade ores are reduced in reverberatory furnaces. Antimony does not react with hydrochloric acid (HCl) in the absence of oxygen.Īntimony can be Separated from the crude antimony sulfide by reduction with scrap iron:Īntimony is Separated from the oxide by a carbothermal reduction: The sulphuric acid reaction produces Sulphur dioxide SO 2 gas. 
SbOCl (s) + 2 OH – (aq) + H 2O (l) ⇌ – (aq) + Cl – (aq)Ģ SbOCl (s) + 2 C 4H 6O 6 (aq) ⇌ 2- (aq) + 4 H + (aq) + 2 Cl – (aq) + 2 H 2O (l)Ģ Sb (s) + 3 F 2 (g) → 2 SbF 3 (s) (Antimony (lll) fluoride)Ģ Sb (s) + 3 Cl 2 (g) → 2 SbCl 3 (s) (Antimony (lll) chloride)Ģ Sb (s) + 3 Br 2 (g) → 2 SbBr 3 (s) (Antimony (lll) bromide)Ģ Sb (s) + 3 I 2 (g) → 2 SbI 3 (s) (Antimony (lll) iodide)Īntimony (Sb) dissolved in hot concentrated sulphuric acid (H 2SO 4) or nitric acid (HNO 3) and forming a Sb(III) solutions. The Precipitate can be dissolved by alkali: Sb (III) as the tetra chloro complex can react with water and forming the precipitate Antimony oxychloride (SbOCl): Grid parameters: a(Hex)=4.307 Å c(Hex)=11.27 ÅĮlectron affinity: 103.2 kJ/mol Nuclear Properties of Antimony ElementĪntimony reacts with in air and form trioxide antimony:Ĥ Sb (s) + 3 O 2 (g) → 2 Sb 2O 3 (s) (Flame is Bluish white)Īt red heat, Reacts with water and form Antimony (lll) oxide: Sb reacts slowly at ambient temperature.Ģ Sb (s) + 3 H 2O (g) → Sb 2O 3 (s) + 3 H 2 (g) The ionization potential of an atom: 8.35 Sound Speed: 3420 m/s Atomic Properties of Antimony Molar magnetic susceptibility: 1.327×10 -9 m 3/mol Physical Properties of Antimonyĭensity: 6.697 g/cm 3 (In solid) 6.53 g/cm 3 (In Liquid) 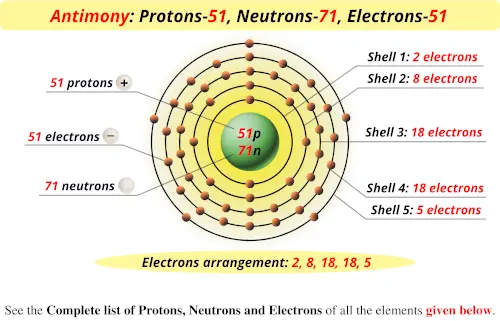
Mass magnetic susceptibility: 10.9×10 -9 m 3/kg Magnetic susceptibility (x mol): -99×10 -6 cm 3/mol Thermal conductivity: 24.4 W/(m∙K) Electrical properties of AntimonyĪ Electrical type: Conductor (poor conductor) Magnetic Properties of Antimony Antimony Electron Configuration Thermal Properties of Antimony 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
